V.C.E Chemistry
Year 11 Revision Notes
Organic Chemistry
Alkenes
– with one substitution
Organic
chemistry is the chemistry of the compounds of carbon.
The
misleading term “organic” is a relic of the days when chemical compounds
were divided into two classes, inorganic and organic, depending upon where they
had come from. Inorganic compounds were those obtained from minerals whereas
organic compounds were those obtained from vegetable or animal sources, that is,
from material produced by living organisms. Indeed, until about 1850 many
chemists believed that organic compounds must have their origins in living
organisms, and consequently could never be synthesised from inorganic material.
These
compounds from organic sources had this in common, they all contained the
element carbon. Even after it had become clear that these compounds do not have
to come from living sources but could be made in the laboratory, it was
convenient to keep the name organic to describe them and compounds like them.
The division between inorganic and organic compounds has been retained to this
day.
Today,
although many compounds of carbon are still most conveniently isolated from
plant and animal sources, most of them are synthesised. They are sometimes
synthesised from inorganic substances like carbonates or cyanides, but more
often from other organic compounds. There are two large reservoirs of organic
material from which simple organic compounds can be obtained, petroleum and
coal. Both of these organic in the old sense, being products of the decay of
plants and animals. These simple compounds are used as the building blocks from
which larger and more complicated compounds can be made.
What
is so special about the compounds of carbon that they should be studied
separately from the compounds of all the other one hundred odd elements of the
Periodic Table?. In part, at least, because there are so many compounds of
carbon, and the compounds of carbon can be so large and complex.
The
number of compounds that contain carbon is many times greater than the number of
compounds that do not contain carbon. These organic compounds have been divided
into families, which generally have no counterparts among inorganic compounds.
Organic
compounds containing thousands of atoms are known, and the arrangement of atoms
in even relatively small molecules can be very complicated. On of the major
problems in organic chemistry is to find out how the atoms are arranged in
molecules, that is, determine the structures of compounds.
There
are many ways in which these complicated molecules can break apart, or rearrange
themselves, to form new compounds and also there are many ways in which atoms
can be added to these molecules, or new atoms substituted for old ones. Much or
organic chemistry is devoted to find out what these reactions are, how they take
place, and how they can be used to synthesise compounds we want.
What
is so special about carbon that it should form so many compounds?. Carbon atoms
can attach themselves to one another to an extent not possible for atoms of any
other element. Carbon atoms can form chains thousands of atoms long, or rings of
all sizes and the chains and rings can have branches and cross links. These
carbon atoms in the rings and chains can have atoms of other elements attached
to them, mainly these are Hydrogen (H), but can also be Fluorine (F), Chlorine (Cl),
Bromine (Br), Iodine (I), Oxygen (O), Nitrogen (N), Sulphur (S) amongst others.
Each
different arrangement of atoms corresponds to a different compound, and each
compound has its own characteristic set of chemical and physical properties.
Organic chemistry is a field of immense importance to technology, it is, for
instance, involved in the chemistry of dyes and drugs, paper and ink, paints and
plastics, petrol and rubber tyres as well as the food we eat and the clothes we
wear.
Methane
CH4
Ethane
CH3 – CH3
(C2H6)
Propane
CH3 – CH2 – CH3
(C3H8)
Butane
CH3 – CH2– CH2 – CH3 (C4H10)
Pentane
CH3 – CH2 – CH2 – CH2 –
CH3
(C5H12)
Hexane
CH3 – CH2 – CH2
– CH2 – CH2 – CH3
(C6H14)
Alkanes
with One Substitution
Bromomethane
CH3 – Br
Chloromethane
CH3 – Cl
Fluoromethane
CH3 – F
Iodomethane
CH3 – I
1-chloro-pentane
CH3 – CH2 – CH2 – CH2 –
CH2– Cl
2-chloro-pentane
CH3 – CH2 – CH2 – CH –
CH3
Cl
3-chloro-pentane
CH3 – CH2 – CH – CH2 –
CH3
Cl
Ethene
CH2 = CH2
(C2H4)
1-Propene
CH3 – CH = CH2
(C3H6)
1-Butene
CH3 – CH2 – CH = CH2
(C4H8)
1-Pentene
CH3 – CH2 – CH2 – CH = CH2 (C5H10)
1-Hexene
CH3 – CH2 – CH2 – CH2 –
CH = CH2
(C6H12)
2-Pentene
CH3 – CH2– CH = CH – CH3
(C5H10)
3-Hexene
CH3 – CH2– CH
= CH – CH2 – CH3
(C6H12)
Chloro-
Ethene CH2 = CH – Cl
Bromo-
Ethene CH2 = CH – Br
Ethyne
CH º
CH (C2H2)
Propyne
CH3 – C º CH (C3H4)
1
– Butyne
CH3 – CH2 –
C º
CH (C4H6)
1
– Pentyne CH3
– CH2 – CH2 – C º
CH (C5H8)
1
– Hexyne CH3
– CH2 – CH2 – CH2 – C
º CH (C6H10)
2
– Hexyne CH3
– CH2 – CH2 – C º
C – CH3
3
– Hexyne CH3
– CH2 – C º
C – CH2 – CH3
Chloro
ethyne CH º C Cl
1
– bromo Pentyne
CH3 – CH2 –
CH2 – C º
C Br
3
– bromo Pentyne CH3
– CH2 – CH2 –
C º
CH
Br
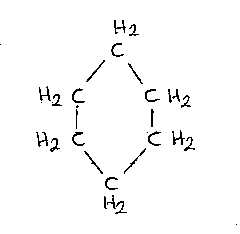
Cyclo
hexane

Benzene
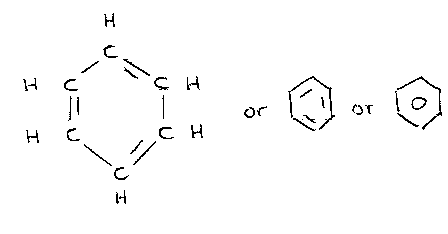
Benzene
C6H6
MethylBenzene C6H5
– CH3
ChloroBenzene C6H5
– Cl
HydroxyBenzene
C6H5 – OH
AminoBenzene
C6H5 – NH2
BenzeneCarboxylic
Acid C6H5 –
COOH
BenzeneCarboxaldehyde
C6H5 – CHO
–
OH
alchohols
–
NH2 amines
–
COOH carboxylic
acids
–
CHO aldehydes
– Cl
chloro
– Br
bromo
– F
fluoro
– I
iodo
– R
alkyls (any alkane,
alkene or alkyne)
Methanol
CH3 – OH
Ethanol
CH3 – CH2 – OH
Propanol
CH3 – CH2 – CH2 – OH
Butanol
CH3 – CH2– CH2 – CH2 –
OH
Pentanol
CH3 – CH2 – CH2 – CH2 –
CH2 – OH
Hexanol
CH3 – CH2 – CH2
– CH2 – CH2 – CH2 – OH
Methanamine
CH3 – NH2
Ethanamine
CH3 – CH2 – NH2
Propanamine
CH3 – CH2 – CH2 – NH2
Butanamine
CH3 – CH2– CH2 – CH2 –
NH2
Pentanamine
CH3 – CH2 – CH2 – CH2 –
CH2 – NH2
Hexanamine
CH3 – CH2 – CH2
– CH2 – CH2 – CH2 – NH2
Methanoic
Acid
H – COOH
Ethanoic
Acid
CH3 – COOH
Propanoic
Acid
CH3 – CH2 – COOH
Butanoic
Acid
CH3 – CH2– CH2 – COOH
Pentanoic
Acid CH3 – CH2 –
CH2 – CH2 – COOH
Hexanoic
Acid CH3 – CH2 –
CH2 – CH2 –
CH2 – COOH
Methanal
H – CHO
Ethanal
CH3 – CHO
Propanal
CH3 – CH2 – CHO
Butanal
CH3 – CH2– CH2 – CHO
Pentanal
CH3 – CH2 – CH2 – CH2 –
CHO
Hexanal
CH3 – CH2 – CH2
– CH2 – CH2 – CHO