V.C.E Chemistry
Year 11 Revision Notes
Gas Laws
Distinguish between solid, liquid and gas in terms of the
Kinetic Molecular Theory.
The Effect of Changes in Temperature and Pressure on
Matter
Solve problems
involving the relationship between temperature, pressure, volume and moles for
gases.
There are four variables used to describe the behaviour
of gases
Dalton's Law of Partial Pressures
Matter
is said to exist in three different physical sates, solid, liquid and gas. The
same substance may exist in all three states under varying conditions such as
temperature and pressure etc.
For
example water may exist as ice (solid), water (liquid) and steam (gas).
The
difference between three states of the same substance can be explained in terms
of the Molecular Kinetic Theory. The basic concepts of this theory are as
follows
·
Molecules
of the gas are in continuous chaotic motion
·
The
molecules move at great speeds, colliding with each other and the container
walls.
·
The
molecules are negligible in size compared with the space between them
·
There
is no attraction or repulsion between the molecules
·
The
average speed of the molecules (their kinetic energy) is directly proportional
to the absolute temperature of the gas.
·
The
molecules in a liquid are moving much slower than the corresponding gas
molecules
·
Molecules
in a liquid are in chaotic motion.
·
Liquid
molecules, due top their reduced speed, are much closer together, thus liquids
are more dense than gases.
·
There
is weak intermolecular forces between molecules in a liquid due to their reduced
speed and denser packing, thus liquids have a fixed volume
·
The
molecules in a solid are moving much more slowly than the corresponding liquid
molecules.
·
Molecules
in a solid are in continuous vibrational motion about a fixed point in the solid
lattice.
·
Molecules
in a solid are close packed.
·
There
are relatively strong intermolecular forces holding the solid molecules into the
solid lattice, thus solids have a fixed volume and shape
Phase
changes are changes in the physical state of a substance
ie.
Melting
Solid
![]() Liquid
Liquid
Boiling
Liquid
![]() Gas
Gas
Condensation
Gas
![]() Liquid
Liquid
Freezing
Liquid
![]() Solid
Solid
Sublimation
Solid
![]() Gas
Gas
Phase
changes can be explained in terms of the Kinetic Molecular Theory as follows
an increase in temperature causes an increase in the
kinetic energy of the molecules with in the solid lattice and the vibrations
eventually overcome the intermolecular forces and the solid lattice breaks down
allowing the particles more mobility
an increase in temperature leads to an increase in the
kinetic energy level of the particles in the liquid. Those particles with
sufficient energy break through the surface of the liquid, overcoming surface
tension) and move into the gas phase. As the temperature increases, eventually
large numbers of particles have sufficient energy to form gas bubbles within the
body of the liquid, and the liquid is said to be boiling. Boiling occurs when
vapour pressure equals external pressure, ie. Air pressure)
by the time the particles in the solid have sufficient
energy to break the intermolecular lattice bonds they also have sufficient
energy to form the gaseous phase of the substance and particles break free into
the gaseous state.
the melting
point of a solid will not be greatly affected by changes in pressure and
providing the substance is pure, is relatively constant.
Some slight effects may be experienced where the solid state is less
dense than the liquid state, e.g. water /
ice where an increase in pressure lowers the melting point.
at normal atmospheric pressure (~ 1 atm) the boiling
points of liquids are relatively
constant. Variations in pressure however cause variations in the boiling points
of liquids. Liquids are said to boil when the internal pressure is equal to the
pressure of the surface of the liquid. So increasing the external pressure
increases the boiling point and decreasing the external pressure decreases the
boiling point.
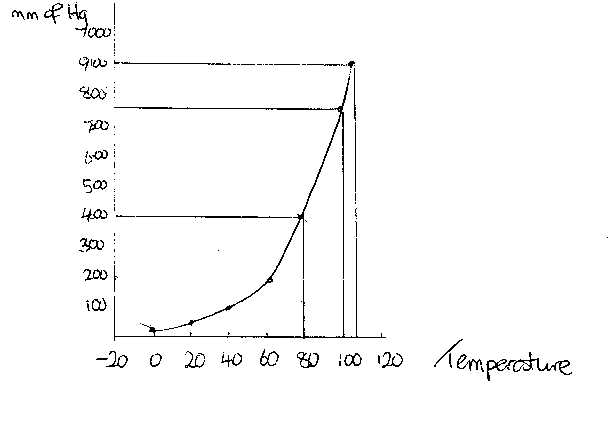
It
can be seen from the above graph that the vapour pressure of liquids increases
with an increase in temperature. The average Kinetic Energy of all particles
increases with an increase in temperature and so at higher temperatures more
particles will “break free” and the vapour pressure will increase.
Solids
also possess a vapour pressure as some
particles can break free of the solid lattice and move into the gaseous state.
Vapour pressures of solids vary greatly from solid to solid. Substances such as
napthalene (moth balls), camphor and iodine have relatively high vapour
pressures compared with iron and glass. The vapour of solids increases with
increase in temperature. The fact that solids possess a
vapour pressure is of great interest in many fields such as freeze drying
of biological samples, blood treatments in blood banks and domestic deodorants
etc.
Pressure
(p)
Volume
(V)
Absolute
Temperature (T)
Number
of moles (n)
The
volume of a given mass of gas is inversely proportional to the pressure (for a
constant temperature)
1
p µ --
(n and T are constant)
V
pV = k (constant)
The
volume of a given mass of gas is directly proportional to the absolute
temperature (for a given pressure)
i.e.
V µ
T (n and p are
constant)
![]() (constant)
(constant)
Extrapolation
of Charles Law to low temperature suggests that there is an absolute lower limit
of temperature. This is supported by the Kinetic Molecular Theory, which
indicates that at – 273° C, molecules would have zero kinetic energy and there
for could not be cooled further. The absolute scale of temperature takes –273°C as its zero and the magnitude of a degree is the same
as on the celcius scale.
So
°K =
°C
+ 273
Combining
Boyles and Charles laws yields
![]() for a given mass of gas , ie
a constant number of moles)
for a given mass of gas , ie
a constant number of moles)
And
this is known as the General Gas Equation
.
k is constant for any given gas at a given mass
The
gas equation can be rewritten
.
pV = nRT
p = atm V = m3
T = °K n = no. of
moles
R is called the Gas Constant and has the value 8.314 JK-1 mole-1
The
molar volume of a gas is the volume occupied by one mole of that gas, which at
STP is 22.4dm3 or 22400 cm3. If a volume of gas V has a
molar volume Vm at the same temperature then
Amount
of Substance (no. of moles) =
![]()
For
gases, equal numbers of gas molecules occupy equal volumes at the same
temperature and pressure
In
a mixture of gases the total pressure of the gases is equal to the sum of the
partial pressures of each gas in the mixture, providing the gases do not react
chemically
..
so PT = P1
+ P2 + P3 + ……….+Pn