V.C.E Chemistry
Year 11 Revision Notes
Structure of Solids
Relate the structure of solids to their Physical
properties.
Crystals
such as NaCl are hard and brittle. They have a high melting point (801C) and are
very poor conductors of electricity in the solid state, but conduct electricity
well in the molten state.

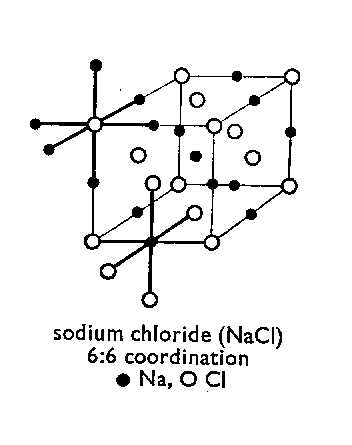
The
lines in the diagram help to show the arrangement of the ions in the lattice,
and must not be taken as chemical bonds. The number of atoms in a tiny crystal
of NaCl is enormous and the diagram should be imagined as extending in all three
directions. It should be noted that each ion is bonded by six ions of opposite
charge. This structure of NaCl is consistent with the observed hardness of the
crystal (ie showing each ions surrounded by six strongly attracted oppositely
charged ions).
The
brittleness of NaCl is also consistent with the diagram of its structure. So if
a deforming force acts on a crystal of sodium chloride and the ions are made to
slide past one another, strong repulsive forces come into play and these forces
rupture the crystal, eg
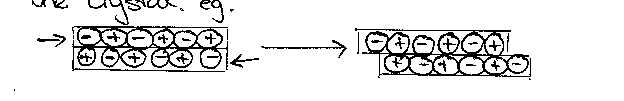
The
structures above suggest that the ions are stationary in the crystal but this is
no so. They are in a state of constant vibration. If the temperature is
increased, the ions vibrate more violently. If the temperature is increased the
ions vibrate more violently, because the additional heat energy absorbed
increases the kinetic energy of vibration in the ions. If the temperature
becomes high enough, the vibrations will become so violent that the crystal
lattice will be destroyed. So even though oppositely charged ions still attract
one another, they are free enough to slide past each other, ie the solid melts.
When the forces between particles comprising a solid are strong, considerable
energy will be needed to disrupt the crystal lattice and melt the solid. So the
stronger he force holding the particles of a substance in the crystal lattice,
the higher the melting point.
These
solids have the following properties
i)
non
conductors of electricity in solid and liquid states
ii)
high
melting and boiling points
iii)
insoluble
in water and most other solvents
iv)
they
are chemically inert
v)
extremely
hard
vi)
brittle
Clearly,
the forces holding the particles of these substances together are extremely
strong. Since the substances are non-conductors in the liquid state the
constituent particles must be uncharged, so that the ionic model is
inappropriate to explain the observed properties. Common examples of these
solids are diamond, silicon dioxide, silicon carbide and tungsten carbide.
In
the case of the diamond lattice it can be seen that each carbon atom is
surrounded by four others (tetrahedral arrangement). The structure consists of a
tetrahedral extending in all dimensions, which suggests covalent bonding between
adjacent atoms. This in turn explains the observed properties of diamond. The
strength of the bonding and the fact that it exists throughout the crystal
accounts for the extreme hardness and high sublimation temperature of diamond.
(as well as the high melting and boiling points of these solids in general).
This is quite understandable since boiling, melting and or sublimation, requires
the rupturing of covalent bonds and very high temperatures are required to do
this.
The
brittleness of diamond is explained by the directional nature of the covalent
bonds, which makes it possible for the atoms to move relative to one another
without the attractive forces between them being drastically weakened.
Consequently the crystal is unable to deform, but shatters if subjected to too
great a force.
The
physical properties of metals include the following
i)
metals
are usually good conductors of heat and electricity
ii)
most
metals are malleable, (can be hammered into sheets) and ductile (can be drawn
into wire)
iii)
their
hardness ranges from soft to very hard (Na is so soft it can be cut with a knife
whereas Cr is very hard
iv)
their
densities range from very high to low, although the densities are usually fairly
high
v)
they
are lustrous (shiny surface) although most metals tarnish and must be scraped to
show lustre
vi)
melting
points range from low to high e.g. Hg (–39°C) to W (3870°C)
In
discussing the structure of metallic solids, sodium will be examined. It is
generally believed that metal consist of a lattice of positive ions through
which moves a cloud of electrons. These electrons are the valency electrons of
the metal and for sodium are the outermost electron from each atom. The positive
ions tends to repel each other however they held together by the cloud of
negatively charged electrons. All the valency electrons move freely throughout
the whole of the crystal lattice and so bind the whole set of particles into a
single unit. A particular valency electron does not belong to any particular
ion, but belongs to the whole crystal.
Electric
current in a metal can be shown to consist of a flow of electrons. If an
electrical potential is applied to the ends of a metallic wire, a general
movement of the electrons the wires is superimposed on their random movement
within the lattice. This results in a number of electrons being forced into one
end of the wire while a similar number of electrons are forced out the other
end. So the electric current passes along the wire. Heat energy can also be
carried by the mobile electrons.
The
positive ions in a metal are not held by rigid bonds but are capable of sliding
past one another if the metal is deformed. This rearrangement of the ions
doesn’t alter the general attractive force in any significant way, so the
metal doesn’t shatter. So metals are generally not brittle.
The
bonding forces can vary widely in metals and this explains the wide variation in
the hardness of metals.
This
wide variation in the bonding forces also accounts for the wide range of melting
points in metals.
The
physical properties of molecular solids include
i)
do
not conduct electricity
ii)
have
low melting points
iii)
they
are quite soft
The
crystals of many organic substances differ strikingly from salts such as NaCl.
Napthalene is a good example of such a compond. Unlike NaCl, the crystals are
soft and have a low melting point (~80°C), although like NaCl,
naphthalene does not conduct electricity in the solid state. These properties
are shared by some inorganic compounds such as hydrogen chloride, oxygen and
carbon dioxide in the solid state. The atoms in all of these compounds are held
together by covalent bonds. These latter substances are gases under normal room
temperatures. All of these substances cann be obtained in the solid state if the
temperature is sufficiently low, and this suggests that there is some force
tending to hold the molecules together. The fact that these solids are very
easily melted and soft suggests that the forces are weak. These intermolecular
forces are called Van der Waals forces, after the Dutch physicist who suggested
their existence. Weak intermolecular forces are also called dispersion forces.
Allotropes
have different structural forms of the one element. Some elements have the
ability to bond covalently in more than one structural form.
The
different structural forms of the element will have different physical
properties such as hardness, density, electrical conductivity, melting point,
and boiling point. However being of identical atoms, the different allotropes of
the one element will form the same compounds and usually undergo similar
chemical reactions. Elements forming allotropes include sulphur, carbon,
phosphorus, tin and oxygen. Carbon will be investigated in depth
Diamonds
occur naturally or they can be made synthetically by subjecting carbon
containing compounds or graphite to very high temperatures and pressures.
Synthetic diamonds are quite tiny but are indistinguishable from naturally
occurring diamond.

Diamond
is one of the hardest substances known. It sublimes at temperatures above 3500°C and is a non conductor of electricity.
One
of the properties of diamond that makes it a highly prized gemstone is its
property of diffracting light to a very marked extent. The cutting of faces of
diamond is designed to use this property to give the gem the greatest possible
amount of “fire”.
Diamond
is the densest form of carbon and its relatively high density (3.5 g cm-3)
reflects the compact nature of the substance. Diamond is extremely resistant to
chemical attack, but it will burn in oxygen if heated to 800°C
The
industrial uses of diamond depend on its great hardness. Black diamonds are used
in rock drills and chips of diamond are used in glass cutters. Industrial
diamonds are also used as abrasives for grinding very hard materials.
Diamond’s non conductivity can be attributed to all its electrons being used
in bonding.
Graphite
occurs naturally in considerable quantities. It is also manufactured in large
quantities in electric furnaces. Coke is packed into furnaces together with a
little iron (III) oxide as catalyst and then covered with a mixture of sand and
coke. The mixture is heated to a temperature of about 3000°C for about 24 hours. Impurities in the coke vapourize
and the product is practically pure graphite.
Physical
Properties of Graphite
Graphite
is a crystalline solid. It has a soft greasy feeling and leaves dark streaks if
it is rubbed on paper. Examination of these black streaks shows that they
consist of a thin layer minute crystals. Graphite is less dense than diamond,
and has a density of 2.2 g cm-3. It has a metallic lustre and is a
fairly good conductor of electricity.
The
structure of graphite has been determined by x ray crystallography, in the same
way as diamond and the structure is shown below.

The
structure shows that graphite consists of layers. The layers are parallel to one
another and there is a relatively wide separation between adjacent layers.
At
least three of the four valence electrons of each carbon atom are used in
forming bonds with the three adjacent carbon atoms in the layer. The fourth
valency electron of each carbon atom is not located by bond formation. The
electrical conductivity of graphite can be explained on the basis of the
mobility of these electrons. The separate layers are too widely spaced to be
bonded together by covalent bonds and are probably bound together by weak Van
der Waals forces which explains why graphite is so soft and flaky.
Graphite
is somewhat more reactive than diamond. Thus is burns more readily and is
attached by nitric acid and sulphuric acid.
Uses
of Graphite
Graphite
is used to make “Lead” pencils. The graphite is powdered and mixed with
clay, that hardness of the “lead” depending on the properties of the clay.
Graphite is a highly refractory material with a melting point of ~ 3500°C. It is widely used for making crucibles to hold molten
metals.
The
electrical conductivity of graphite makes it suitable as the electrode material
in electrical furnaces and arc lights. In addition, graphite is not attacked by
chlorine and its is therefore used for electrodes during electrolysis, if
chlorine is evolved at the electrode.
Graphite
is also a good lubricant, because adjacent layers slide easily over one another.
It is particularly useful for lubricating moving parts which are subject to high
temperatures, where mineral oils would burn away.