V.C.E Chemistry
Year 11 Revision Notes
Acids and Bases
Define an acid as a proton donor.
Define a base as a Proton acceptor.
Given relevant information, select acid and/or base.
Acid-Base Reactions in Aqueous Solution
Distinguish between strong and weak acids on the basis of
completeness of ionization.
Distinguish between strong and weak bases on the basis of
completeness and ionization.
Distinguish between acidic. basic and neutral solutions
using the pH system.
Calculate pH from [H+] or [OH-]
Calculating the pH of a Solution
Measurement of pH of a Solution
Determine the molarity and concentration of common strong
acids and common strong bases.
State why aqueous solutions of only some salts of
neutralization reactions have a pH = 7
Name all the Ions and Molecules which Aqueous Solutions
in CO2, SO2, NH3
An
acid is a chemical species capable of donating a proton e.g. HCl, H2SO4,
CH3COOH, H2CO3, H3PO4, HNO3
A
base is a chemical species capable of accepting a proton, e.g. NaOH, NH3.
KOH
The
reaction of an acid with a base is therefore characterised by the transfer of a
proton from the acid to the base. Consider for example the hydrolysis of acetic
acid. IN this reaction, CH3COOH donates a proton to water, this
indicates that CH3COOH is an acid in the Bronsted sense and H2O
functions as a base.
------- H+ -----à
CH3COOH
+ H2O
![]() CH3COO-
+ H3O+
CH3COO-
+ H3O+
Acid base
i)
HA +
B
![]() BH+ +
A-
BH+ +
A-
acid 1 base 1
acid 2
base 2
e.g.
HCl
+ NH3
![]() Cl- +
NH4+
Cl- +
NH4+
acid
1 base 1
base 2
acid 2
NOTE
These
reactions are shown as reversible and in the reverse reaction, acid 2 acts as
the acid donating the proton.
i.e.
HCl and NH4+ are both acids (HCl is a much stronger acid
and the reaction is mainly forward) and NH3 and Cl-
are both bases. HCl and Cl-
differ only by one proton and they are called a conjugate pair. So, Cl-
is the conjugate base of the acid HCl. Similarly, NH3 and NH4+
are also a conjugate pair with NH4+ being the
conjugate acid of the base NH3
Water
may react either as an acid by forming OH- or as a base by forming H3O+.
e.g.
i)
as an acid
H2O
+ NH3
![]() NH4+
+ OH-
NH4+
+ OH-
Acid
conj base
ii)
as a base
H2O
+ HCl
![]() H3O+
+ Cl-
H3O+
+ Cl-
Base
conj
acid
Water
can also react with itself, but only very slightly
-----(-)--------à
H2O
+ H2O
![]() H3O+
+ OH-
H3O+
+ OH-
Acid
1 base
2
acid 2 base 1
Another
example of such a substance is HCO3-
As
a base
HCO3-
+ H2O
![]() H2CO3
+ OH-
H2CO3
+ OH-
As
an acid
HCO3-
+ H2O
![]() CO3
2- +
H3O+
CO3
2- +
H3O+
Entities
that can act as both an acid and a base are called amphiprotic
Some
molecules can donate more than one proton. An example would be sulphuric acid,
H2SO4
i.e.
H2SO4
+ H2O
![]() H3O+
+ HSO4 -
H3O+
+ HSO4 -
HSO4
- +
H2O
![]() H3O+
+ SO42-
(incomplete)
H3O+
+ SO42-
(incomplete)
The
sulphuric acid molecule donates two protons to the base, H2O.
Sulphuric acid is called a diprotic acid.
In
a similar way phosphoric acid can donate protons,
i.e.
H3PO4
+ H2O
![]() H3O+
+ H2PO4 -
(partial)
H3O+
+ H2PO4 -
(partial)
H2PO4
- +
H2O
![]() H3O+
+ HPO4 2 - (slight)
H3O+
+ HPO4 2 - (slight)
HPO4
2 - +
H2O
![]() H3O+
+ PO4 3 -
(very slight)
H3O+
+ PO4 3 -
(very slight)
Such
an acid is said to be triprotic
The
strength of an acid depends on the degree to which it ionises in aqueous
solution. Strong acids such as HCl, HNO3 , H2SO4
ionize virtually to completion whereas weak acids such as CH3COOH,
H2CO3 and H3PO4
ionize only partly or
very little at all. Sulphurous acid, H2SO3 can be referred
to as an acid with moderate strength. It is a stronger than the weaker acids
listed here, but it is not as strong at the stronger acids mentioned.
The
strength of am acid is also greatly influenced by the relative strength and
stability of its conjugate base. Strong acids generally have very stable
conjugate bases, which are consequently weak bases. The reverse reaction is
there for negligible, e.g, Cl- is a stable and weak base. Weak acids
relatively unstable conjugates which are quite strong bases and the reverse
reaction is considerable, so CH3COO- is a strong base
compared to Cl-
In
general strong acids have
weak conjugate bases
weak acids have strong conjugate bases
The
strength of a base primarily depends on its ability to absorb a proton. IN the
case of bases soluble aqueous solutions (alkalis), the strength of the base
depends on the degree of dissociation of the molecule in water. So bases with a
high degree of dissociation in water will be strong bases while those bases with
a low degree of dissociation in water will be weak acids
NaOH
![]() Na+ + OH-
complete à strong base
Na+ + OH-
complete à strong base
KOH
![]() K+ + OH-
complete
à strong base
K+ + OH-
complete
à strong base
Ca(OH)2
![]() Ca
2+ +
2OH-
slight à weak base
Ca
2+ +
2OH-
slight à weak base
Mg(OH)2
![]() Mg
2+ + 2OH-
slight à weak base
Mg
2+ + 2OH-
slight à weak base
NH3
+ H2O
![]() NH4
+ +
OH-
slight à weak base
NH4
+ +
OH-
slight à weak base
To
obtain a numerical system for comparing the strengths of acids and bases a
system has been established, the pH system.
The
concentration of H3O+
ion in solution is taken as the standard. Pure water is taken as
the zero point or point of neutrality, where the concentrations of both H3O+ and OH-
are exactly equal
Note
The greater the concentration of H3O+ , the
stronger is the acidity of the solution.
The
symbol [ ]
indicates the concentration of a solution in molarity
The
[H3O+ ] of 0.1 M HCl
is 10-1
The
[H3O+ ] of pure water is 10-7
The
[H3O+ ] of 0.1 M NaOH is 10-13
As
these values of [H3O+ ] are difficult to work with, a
further definition of the acidity of a solution is made – the pH of a solution
So
pH = – log10 [H3O+
]
So
0.1 M HCl, pH = 1 pure
water, pH =7 0.1 NaOH, pH = 13
Distinguishing
between acid, basic and neutral solutions using the pH system becomes quite a
simple matter.
1)
If
the pH of a solution is less than 7, the solution is acidic, the lower the pH of
the solution, the stronger the acidity
2)
If
the pH of a solution is equal to 7, the solution is neutral [OH- ]
= [H3O+ ] = 10-7
3)
If
the pH of a solution is greater than 7, the solution is basic, the higher the pH
of a solution, the stronger the bascisity
In
pure water
2
H2O
![]() H3O+
+
OH-
H3O+
+
OH-
[H3O+
] = [OH- ]
= 10-7 (pH = 7)
[H3O+ ]
X [OH- ]
= 10-14
so
pH + pOH =
14
To
determine the pH of a solution knowing it’s [H3O+ ]
is simple : pH = – log10 [H3O+ ]
So
example : calculate the pH of a solution of 0.01M HCl
\ pH = –
log10 (0.01)
= – log10 (10-2)
= 2
To
determine the pH of a solution knowing its [OH- ] is also quite simple, but it requires 2 steps.
So
Example : calculate the pH of a
solution of 0.01 M NaOH
\ pOH = –
log10 (0.01)
= – log10 (10-2)
pOH = 2
since
pH + pOH = 14
pH = 14 – pOH
= 14 – 2
pH = 12
There
are three basic methods of measuring the pH of a solution
this
is specially treated paper which will change colour as soon a sit is placed in a
solution containing H3O+. Depending on the colour change
the pH of the solution can be determined approximately. Using pH paper has the
benefit of being quick and easy, however, it is only an approximate value of the
pH of the solution (~ 1 to 0.5 of a pH unit) and each piece of paper can only be
used for one measurement.
this
is an electronic device which will give a quick and very accurate reading of the
pH of a solution containing H3O+ , as soon as the electrodes connected to the meter are placed
in the solution. Using a pH meter has many advantages, it is quick, accurate
provided the machine and its electrodes are calibrated correctly and cared for
will last for many years of service
during
an acid / base titration we want to know exactly when the reaction between the
acid and the base is finished, i.e. when the acid and the base have just
neutralized on another. To do this we use acid / base indicators, which are
substances that undergo definite colour changes within a fairly narrow range of
pH. That is, the indicators change colour just as soon as the reaction between
the acid and the base is finished
|
Indicator |
Ph
Range for Colour
Change |
Colour
at Lower pH |
Colour
at Higher pH |
|
Thymol
Blue |
1.2
– 2.8 |
Red |
Yellow |
|
Methyl
Orange |
3.1
– 4.4 |
Red |
Yellow |
|
Methyl
Red |
4.2
– 6.3 |
Red |
Yellow |
|
Litmus |
5
– 9 |
Red |
Blue |
|
Bromothymol
Blue |
6.0
– 7.6 |
Yellow |
Blue |
|
Thymol
Blue |
8.0
– 9.6 |
Yellow |
Blue |
|
Phenylphthalon |
8.3
– 10.0 |
Colourless |
Red |
A
universal indicator is really a mixture of several indicators chosen to display
a variety of colours over a particular range of pH. Universal indicators are
useful in the determination of the approximate pH of solutions.
Neutralization
Reactions of Acids and Bases
In
general, when an acid and a base react with one another, the products are water
and a salt
e.g.
HCl
+ NaOH
![]() NaCl
+ H2O
NaCl
+ H2O
Acid
base
salt
H2SO4
+ 2KOH
![]() K2SO4
+ 2 H2O
K2SO4
+ 2 H2O
Acid
base
salt
Equations
such as these in which no charged particles (ions) are shown are called
molecular equations for the neutralization of an acid with a base.
When
equations in which charged particles (ions) are used, the equations are called
ionic equations for the neutralization of an acid with a base.
e.g.
NaOH(s)
+ (aq)
![]() Na+
(aq) + OH- (aq)
Na+
(aq) + OH- (aq)
HCl
(l) + (aq)
![]() H+
(aq) + Cl- (aq)
H+
(aq) + Cl- (aq)
Ionic
equation H+ (aq)
+ OH- (aq)
![]() H2O
H2O
Example
1 A 20ml aliquot of 1.00M NaOH is
titrated to end point with 15.2ml of a H2SO4 solution of
unknown concentration. Calculate the molarity of the H2SO4
solution.
H2SO4
+ 2NaOH
![]() 2
H2O + Na2SO4
2
H2O + Na2SO4
1
mole
2 moles
x
mole
2x moles
no.
of moles of NaOH present =
![]()
= 0.02 mole
\ no. of mole
of H2SO4 present is 0.01
(
![]() )
)
\ in 15.2 ml of H2SO4, there are
0.01 moles of H2SO4
\ the molarity of the H2SO4 solution
=
![]() X 0.01
X 0.01
=
0.66M
example
2 A 20ml aliquot of a 0.5M H2SO4 solution is titrated to
end point with 25 ml of a KOH solution of unknown concentration. Calculate the
molarity of the KOH solution
H2SO4
+ 2 KOH
![]() 2
H2O + Na2SO4
2
H2O + Na2SO4
1
mole 2 mole
in
20 ml of 0.5M H2SO4 solution there are
![]() =
0.01 moles of H2SO4
=
0.01 moles of H2SO4
\ in 25ml of
KOH solution there are 0.02 moles of KOH
(0.01 X 2)
\ the
molarity of KOH solution is
![]() = 0.8M
= 0.8M
b.
Experimentally determine the concentration of a common strong acid or
base using a common strong base or acid.
Only
some aqueous solutions of salts of neutralization have a pH = 7. Normal salts do
not have a replaceable H+, so they cannot donate H+
The
aqueous solutions of salts of neutralization reactions resulting from the
neutralization of a strong acid and a strong base are neutral, i.e. pH = 7
The
aqueous solutions of salts of neutralization reactions resulting from the
neutralization of a strong acid and a weak base are acidic, i.e. pH < 7
The
aqueous solutions of salts of neutralization reactions resulting from the
neutralization of a weak acid and a strong base are basic, i.e. pH > 7
The
pH of the solution of aqueous solutions of salts of neutralization treactions
varies greatly in the area of the equivalance or end point
Example
Titration of NaOH with HCl
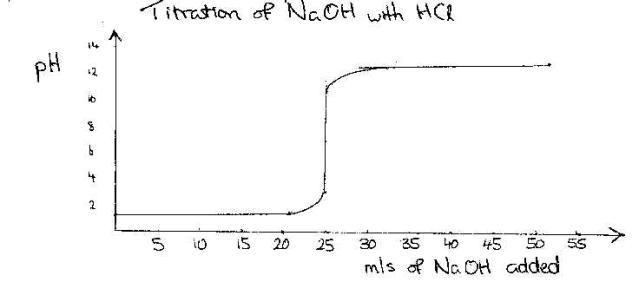
This
occurs because in the vicinity of the end point (where the acid and base have
nearly neutralized one another) the addition of very minute volumes of titrant
(NaOH) caused large changes in the pH of the solution.
Because
of the fact that aqueous solutions of only some salts of neutralization
reactions have a pH = 7, a wide variety of acid / base indicators are required,
to show when a certain acid / base has reached its equivalence point.
The
table of acid / base indicators mentioned earlier showing the pH range in which
they work is often referred to.
i)
CO2 +
H2O
![]() H2CO3
(Carbonic Acid)
H2CO3
(Carbonic Acid)
This
reaction is only slight, about 1%
Carbonic
acid is only a weak acid. The evidence for this lies in its poor conductivity
and is supported by the litmus test. Carbonic Acid is only slightly ionised in
the excess water.
H2CO3
+ H2O
![]() H3O+
+ HCO3–
(slight)
H3O+
+ HCO3–
(slight)
Hydronium Hydrogen
Carbonate
Ion
Ion
The
hydrogen carbonate ion is capable of acting as an acid, although it is a much
weaker acid than carbonic acid.
HCO3-
+ H2O
![]() H3O+
+ CO32–
(very slight)
H3O+
+ CO32–
(very slight)
ii)
SO2 +
H2O
![]() H2SO3
(Sulphurous Acid)
H2SO3
(Sulphurous Acid)
The
solution formed is only weakly acidic and is probably similar to carbon dioxide
solution in that it contains molecules of the gas which are dissolved but have
not reacted with the water. Sulphurous acid will ionise only to a slight extent
in the excess water.
H2SO3
+
H2O
![]() HSO3
–
+ H3O+
HSO3
–
+ H3O+
Hydrogen Sulphite
Hydronium
Ion
Ion
Further
ionisation of the HSO3 – ions will occur but only to a very slight extent.
HSO3
–
+ H2O
![]() SO3
2–
+ H3O+
SO3
2–
+ H3O+
Sulphite
Hydronium
Ion
Ion
The whole solution is called sulphurous acid, and because it contains several different molecules and ions, it cannot be simply represented by any one formula. However as a matter pf convenience, the formula H2SO3 is usually used to represent the solution.
iii)
NH3 +
H2O
![]() NH4+
+ OH-
NH4+
+ OH-
Ammonium Hydroxide
Ion
Ion
A
solution of ammonia inn water is alkaline to litmus. The solution is a poor
conductor of electricity and so it can be said, it contains very few ions.