V.C.E Chemistry
Year 11 Revision Notes
Oxidation and Reduction (REDOX)
Reduction and Oxidation
(REDOX)
Use a given Electrochemical Series to determine which
reactions will occur
show the direction of the electron flow in the external
circuit;
Ion Movement in the Electrolyte
predict what will happen at the electrodes.
Reactions of Metals with Water
Tin, Lead, Copper, Mercury and Silver
Reactions of Metals with Hydrochloric Acid
Electroplating Metals With Copper
Production and Purification of Substances
why corrosion of iron takes place
Mechanism of Corrosion of Iron
Oxidation
and reduction reactions occur simultaneously
can
be defined as the removal of electrons from a species
e.g.
Fe 2+
![]() Fe
3+ + e –
Fe
3+ + e –
Cu +
![]() Cu
2+ + e –
Cu
2+ + e –
Sn 2+
![]() Sn
4+ + e –
Sn
4+ + e –
can
be defined as the addition of electrons to a species
e.g.
Fe 3+ + e –
![]() Fe
2+
Fe
2+
Cu 2+ +
e –
![]() Cu
+
Cu
+
Sn 4+ +
e –
![]() Sn
2+
Sn
2+
It
is common to separate Redox reactions into its two component parts which is a
convenient way of indicating which species gains electrons or loses electrons.
Thgese component parts are called HALF REACTIONS. The electrons
symbolized in these equations are cancelled out when te half reactions are
combined.
e.g.
Consider the reaction when zinc is immersed in copper(II) sulphate solution. The
overall chemical reaction is expressed as
Zn +
CuSO4
![]() ZnSO4 + Cu
ZnSO4 + Cu
The
reaction can be further expressed by the equation
Zn +
Cu 2+
![]() Zn
2+ +
Cu
Zn
2+ +
Cu
Zn
is being oxidised to Zn 2+
Cu
2+ is being reduced to
Cu
This
reaction can be resolved into two half reactions. The one depicting oxidation of
zinc
Zn
![]() Zn
2+ + 2 e –
Zn
2+ + 2 e –
And
the other indicating the reduction of copper(II) ions
Cu
2+ + 2 e –
![]() Cu
Cu
The
driving force for the above reaction can be measured by placing a voltage
measuring device in the circuit. We may consider this force as being the sum of
two potentials called half cell potentials or single electrode potentials, one
of these is associated with the half cell reaction occurring at the anode and
the other is associated with the half cell reaction talking place at the
cathode.
We
cannot measure absolute potentials for half cell reactions but the relative half
cell potentials that can be measured are quite useful. To obtain consistent
relative half cell potential data, it is necessary to compare all electrodes
against a common reference. The reference electrode should be easy to construct,
exhibit reversible behaviour, and give constant and reproducable potentials for
a given set of experimental conditions. The standard hydrogen electrode (S.H.E.)
meets these requirements and is universally used as the ultimate reference
electrode. The electrode basically consists of a platinum wire immersed in a
solution containing hydrogen ions, and hydrogen gas is bubbled across the
surface of the platinum. This type of electrode is called a gas electrode
because the platinum takes no part in the electrochemical reaction. The half
cell reaction of the cell is given as
H2
(g)
![]() 2H+
+ 2 e –
2H+
+ 2 e –
By
definition this reaction is said to have a zero potential or an E° of zero or E° = 0. By convention, when quoting E° values, the half reactions are
written as reductions.
is
a list of half reactions written in descending order according to their E° values
|
Half
Reaction Oxidizer
+ electrons
|
Standard Electrode Potential
(volts) |
|
F2 + 2e –
|
+2.87 |
|
H2O2 + 2H+ + 2e –
|
+1.77 |
|
Mn04– + 8H+ + 5e –
|
+1.52 |
|
Cr2072– + 14H+ + 6e–
|
+1.36 |
|
Cl2 + 2e –
|
+1.36 |
|
Mn02 + 4H+ + 2e –
|
+1.28 |
|
Br2 + 2e –
|
+1.07 |
|
HN02 + H+
+ e –
|
+0.99 |
|
NO3–
+ 3 H+ +
2e –
|
+0.94 |
|
Hg 2+ + 2e –
|
+0.85 |
|
Ag+ + e –
|
+0.80 |
|
Fe 3+
+ e –
|
+0.77 |
|
02 + 2 H+
+ 2e –
|
+0.68 |
|
I2 + 2e –
|
+0.54 |
|
Cu2+ + 2e –
|
+0.35 |
|
S042– +
4 H+ + 2e–
|
+0.20 |
|
S + 2H+
+ 2e –
|
+0.14 |
|
2H+ + 2e –
|
0.00 assigned |
|
Pb2+ + 2e –
|
–0.12 |
|
Sn 2+ + 2e –
|
–0.14 |
|
Ni 2+ + 2e –
|
–0.25 |
|
Fe 2+ + 2e –
|
–0.44 |
|
2C02 + 2 H+
+ 2e –
|
–0.49 |
|
Cr 3+ + 3e –
|
–0.71 |
|
Zn 2+ + 2e –
|
–0.76 |
|
Mn 2+ + 2e –
|
–1.05 |
|
Al 3+ + 3e –
|
–1.67 |
|
Mg 2+ + 2e –
|
–2.34 |
|
Na+ + e –
|
–2.71 |
|
Ca 2 + + 2e –
|
–2.87 |
|
Ba 2+ + 2e –
|
–2.90 |
|
K+ + e –
|
–2.92 |
–
Using
some degree of caution, this electrochemical series can be used to predict which
chemical reaction will take place when two chemicals are mixed.
So
if we consider the reaction of zinc immersed in copper sulphate again
From
the table
The
Cu2+ / Cu electrode has a value of +0.35V
And
the Zn2+ / Zn electrode has a value of
–0.76V
It
can be said, using these values, that the Cu2+ will reduce to Cu and
the Zn will oxidize to Zn2+
(14)
Given a diagram or description electrolytic or galvanic cell:
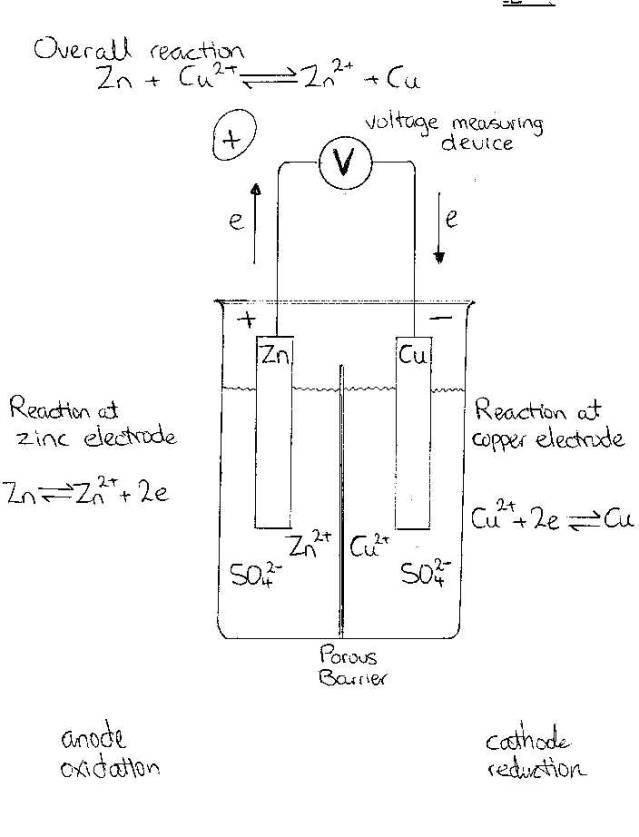
A
cell consists of a pair of conductors or electrodes, usually metallic, each of
which is immersed in an electrolyte. When the electrodes are connected by an
external conductor and a dlow of electrons occurs, a chemical oxidation occurs
at the surface of one electrode and a reduction occurs at the surface of the
other.
Well
a cell is operated to produce electric energy, it is called a Galvanic or
sometimes Voltaic cell. A cell requiring an external source of electric energy
is called an electrolytic cell
Referring
to the diagram of The Electrochemical cell above. This cell is a galvanic cell
because when the two electrodes are connected by a wire, electric energy is
produced, and a flow of electrons from the zinc electrode to the copper occurs.
This
cell can operate as an electrolytic cell if a battery is introduced into the
external circuit, which would force electrons to flow in the opposite direction
through the cell. In this case Zinc would deposit and copper would dissolve,
consuming energy from the battery.
By
definition
The
Anode is the electrode at which oxidation occurs in the both the electrolytic
and galvanic cells. It has a positive charge
The
Cathode is the electrode at which reduction occurs in the both the electrolytic
and galvanic cells. It has a negative charge .
So
in the diagram,
Oxidation
is occurring at the Zinc electrode so this is called the Anode
Reduction
is occurring at the Copper electrode so this is called the Cathode
The
electrons flow from anode to cathode
;
We
have seen that the electrode reactions occurring in the galvanic cell studied
above are
Zn
![]() Zn
2+ + 2 e –
Zn
2+ + 2 e –
Cu
2+ + 2 e –
![]() Cu
Cu
As
a result, the solution in the cell compartment containing the zinc electrode
shows an increase in the concentration of zinc ions, while the solution
surrounding the copper electrode is depleted of copper (II) ions. If no interna;
contact existed between the two parts of the cell, a charge imbalance would
develop in the neighbourhood of the electrodes, e.g. an excess of positive ions
would be found near the zinc electrode and an excess of negative ions near the
copper electrode.
As
a result of this charge imbalance, no current would flow. With the arrangement
shown in the diagram, direct contact between the metallic zinc and copper (II)
ions is prevented by the porous barrier, but it does not permit the passage of
ions.
As
a result, zinc ions as well as other cations (positive ions), can migrate from
the solution surrounding the zinc electrode toward the copper electrode. Anions
(negative ions) can also migrate, but in the opposite direction, that is anions
can migrate from the solution surrounding the copper electrode toward the zinc
electrode.
The
passage of current through a cell involves
the migration of ions within the solution, and the current may be considered to
be carried by these ions. Not only the ions that react at the electrodes but all
ions present in the solution, participate in the carrying of the current,
The
following points should be noted
1)
Electrons
always flow from the anode to the cathode
2)
Oxidation
always occurs at the Anode
3)
Reduction
always occurs at the Cathode
Na
![]() Na+ +
e –
X2 oxidation
Na+ +
e –
X2 oxidation
2H2O
+ 2 e –
![]() 2OH– + H2
reduction
2OH– + H2
reduction
----------------------------------------------
2Na + 2H2O
![]() 2
Na+ + 2OH–
+ H2
2
Na+ + 2OH–
+ H2
reducer
oxidiser
K
![]() Na+ +
e –
X2 oxidation
Na+ +
e –
X2 oxidation
2H2O
+ 2 e –
![]() 2OH– + H2
reduction
2OH– + H2
reduction
----------------------------------------------
2K +
2H2O
![]() 2
K+ +
2OH– +
H2
2
K+ +
2OH– +
H2
reducer
oxidiser
Ca
![]() Ca2+ +
2e –
oxidation
Ca2+ +
2e –
oxidation
2H2O
+ 2 e –
![]() 2OH– + H2
reduction
2OH– + H2
reduction
----------------------------------------------
Ca +
2H2O
![]() Ca2+ +
2OH– +
H2
Ca2+ +
2OH– +
H2
reducer
oxidiser
Similar
reaction to calcium but very much slower in cold water. With hot water, the
reaction is somewhat faster but is still quite slow.
If
reacted with steam, the magnesium burns forming a white powdery residue.
Mg +
H2O
![]() MgO
+ H2
MgO
+ H2
reducer oxidiser
Aluminium
does not react with water. However, if mercury is rubbed over the surface of the
aluminium, the metal slowly displaces hydrogen even from cold water. The lack of
reactivity of aluminium can be attributed to the thin layer of oxide that forms
on the surface of aluminium.
If
stream is passed over zinc in a furnace to 400°C, a reaction will take place
slowly
Zn +
H2O
![]() ZnO
+ H2
ZnO
+ H2
Similar
to zinc but a temperature of about 700°C
is required
3Fe
+ 4 H2O
![]() Fe3O4
+ 4H2
Fe3O4
+ 4H2
Fe3O4
= (Fe2+) (Fe3+)2
(O2–)4
A
reversible sign is used in the reaction equation because the reverse reaction
can occur. So hydrogen is if Hydrogen is passed over hot Fe3O4,
the oxide is reduced to iron and the hydrogen is oxidised to steam.
These
metals do not react with water or steam even at fairly high temperatures.
If
magnesium is placed in dilute hydrochloric acid it dissolves rapidly with
vigorous evolution of a colourless gas which is hydrogen
Mg
![]() Mg2+ +
2e –
oxidation
Mg2+ +
2e –
oxidation
2H+ +
2 e –
![]() H2
reduction
H2
reduction
----------------------------------------------
Mg +
2H+
![]() Mg2+ +
H2
Mg2+ +
H2
reducer
oxidiser
Two
chloride ions will remain unused for each magnesium ion formed. These are
spectator ions and partner the magnesium ion in solution. So the solution
contains magnesium chloride.
Mg
+ 2H+ +
2Cl2–
![]() Mg2+ + 2Cl2–
+
H2
Mg2+ + 2Cl2–
+
H2
At
first there appears to be no reaction but after a while, particularly if the
mixture is warmed, the aluminium
begins to react. The delay to the start of the reaction can be attributed to the
protective layer of oxide on the surface of the aluminium.
Al
![]() Al3+ +
3e – X
2 oxidation
Al3+ +
3e – X
2 oxidation
2H+ +
2 e –
![]() H2
X
3 reduction
H2
X
3 reduction
----------------------------------------------
2Al +
6H+
![]() 2Al3+ +
3H2
2Al3+ +
3H2
reducer
oxidiser
The
reaction is similar to the reaction of magnesium except it is slower. The
hydrogen is produced at a slower rate without frothing and as a result is often
used to prepare hydrogen
Zn
![]() Zn2+ +
3e –
oxidation
Zn2+ +
3e –
oxidation
2H+ +
2 e –
![]() H2
reduction
H2
reduction
----------------------------------------------
Zn +
2H+
![]() Zn2+ +
H2
Zn2+ +
H2
reducer
oxidiser
Iron
is similar to zinc but the reaction is slower and often the acid must be heated
to produce a reasonably rapid evolution of gas
Fe
![]() Fe2+ +
3e –
oxidation
Fe2+ +
3e –
oxidation
2H+ +
2 e –
![]() H2
reduction
H2
reduction
----------------------------------------------
Fe +
2H+
![]() Fe2+ +
H2
Fe2+ +
H2
reducer
oxidiser
Granulated
Tin dissolves very slowly in in cold dilute hydrochloric acid. If the mixture is
heated., or if hot concentrated hydrochloric acid is used, the tin dissolves
more rapidly
Sn
![]() Sn2+ +
3e –
oxidation
Sn2+ +
3e –
oxidation
2H+ +
2 e –
![]() H2
reduction
H2
reduction
----------------------------------------------
Sn +
2H+
![]() Sn2+ +
H2
Sn2+ +
H2
reducer
oxidiser
Lead
slowly becomes coated with a white layer of insoluble lead (II) chloride when
placed in cold hydrochloric acid, which slows down the reaction. However, lead
dissolves fairly rapidly in hot concentrated HCl.
There
is no apparent reaction when these metals are p[laced in hydrochloric acid. Thus
they do not reduce the hydrogen ions in aqueous solution of HCl.
The
sequence of metals arranged in order of the readiness with which they react with
water or steam is the same as their sequence of reactivity with dilute
hydrochloric acid or sulphuric acid and salt solutions. The similarity in
sequence suggests a similarity in there actions of the metals in each case. This
has been illustrated by showing that each reaction can be interpreted as
donations of electrons by atoms of the metals during formation of positive ions.
The sequence is called the activity series or the displacement series of the
metals. It is also the order of ease of formation of positive ions in solution
|
Metal |
Activity with Cold Water |
Activity with Steam |
Activity with Acids |
Activity with Solutions of
Metallic Salts |
|
K |
Displace |
|
|
|
|
Na |
Hydrogen
from |
|
|
|
|
Ca |
Cold
water |
|
|
|
|
Mg |
Do
not displace |
Displace
|
Displace
|
Displace
|
|
Al |
hydrogen |
Hydrogen |
Hydrogen
from |
lower |
|
Zn |
From
cold water |
from
steam |
Hydrochloric
and |
Metals |
|
Fe |
|
|
Dilute
sulphuric |
from |
|
Sn |
Do not displace |
Hydrogen |
acids |
solutions
|
|
Pb |
From cold water |
or steam |
|
of
their |
|
Cu |
Do not displace |
Hydrogen
from |
|
salts |
|
Hg |
cold water, steam |
or
acids |
|
|
|
Ag |
|
|
|
|
This is widely employed to forma coating of copper over a
base metal, prior to plating with other metals, such as nickel or chromium.
The article to be copper plated is first cleaned of rust
or grease and is then made the cathode (negative) in an electrolytic cell. The
electrolyte is copper (II) sulphate solution containing a little sulphuric acid.
The sulphuric acid improves the conductivity of the electrolyte solution and
prevents rough coatings. If firm deposits are to be formed, careful attention
must be given to the concentration of the electrolyte, the current used, the
size of the cathode and the temperature of the bath.
The main source of zinc, is zinc blende, which is found
mixed with large amounts of other minerals, such as lead sulphide (PbS), pyrites
(Fe S2) and silica (SiO2). The ore is finely ground and
the useful components are separated out by selective flotation. The zinc sulfide
is then burnt in air to form zinc oxide and sulfur dioxide
2ZnS
+ 3O2
![]() 2ZnO
+ 2SO2
2ZnO
+ 2SO2
The zinc oxide can be reduced to zinc in one of two ways
i)
reduction
by carbon
A mixture of zinc oxide and carbon (e.g. coke) is
strongly heated
ZnO
+ C
![]() Zn
+ CO
Zn
+ CO
The zinc oxide is dissolved in dilute sulphuric acid
ZnO + 2H+
![]() Zn
2+ + H2O
Zn
2+ + H2O
Zinc dust is then added to the solution to displace all
metals below zinc in the activity series. These may be present from impurities
in the ore.
e.g. Zn
+ Cu 2+
![]() Zn
2+ + Cu (s)
Zn
2+ + Cu (s)
The solution is electrolysed using a lead anode (which is
not attacked by oxygen or acids) and an aluminium cathode. The electrode
reactions are
Cathode Reaction
Zn 2+ +
2e–
![]() Zn
Zn
Anode Reaction
2H2O
![]() 4H+
+ O2 + 4e–
4H+
+ O2 + 4e–
So the zinc ions are replaced by hydrogen ions and the solution contains
sulphuric acid. This is used to dissolve the next batch of zinc oxide. The zinc
is peeled from the aluminium cathodes when the deposit has reached a suitable
thickness
The main source of aluminium is bauxite (Al2O3 . x
H2O). The first stage of the process is the purification of the
aluminium oxide, which is done by dissolving the bauxite in hot concentrated
caustic soda solution under pressure
Al2O3 + 3H2O
+ 2OH–
![]() 2Al(OH)4–
2Al(OH)4–
The
sodium ions partner the aluminate ions formed and the solution formed is a
solution of sodium aluminate.
When
the solution is cooled, diluted and seeded with freshly precipitated aluminium
hydroxide, most of the aluminate changes into aluminium hydroxide, which
precipitates in a coarsely crystalline form and is easily filtered.
2Al(OH)4–
![]() Al(OH)3 +
OH–
Al(OH)3 +
OH–
The
precipitate is filtered off, and is heated to decompose it to aluminium oxide, a
white powder which is also called alumina
Al(OH)3
![]() Al2O3
+ 3H2O
Al2O3
+ 3H2O
The
alumina is dissolved in a molten mineral called cryolite (Na3AlF6)
producing an electrolytic conductor. Aluminium is formed at the cathode of a
complicated cell.
The most usual shape is a cylinder with a carbon rod down
the centre. This is surrounded by a solid mixture of MnO2(s), NH4Cl
(s) and C(s). The mixture is wet with an electrolyte
consisting of a solution containing
ZnCl2 and NH4Cl. Because NH4+ is
an acid, the electrolyte solution is acidic. The outer container of the
cylinder is made of zinc.
The electricity produced by the cell is generated by
chemical reactions
Of the Zn and MnO2. The Zn dissolves and
releases electrons and hence generates a negative charge on the zinc container.
Zn
![]() Zn2+
+ 2e–
Zn2+
+ 2e–
The MnO2 consumes H+ ions from the electrolyte and electrons
from the carbon rod. Hence a
positive charge is generated on the carbon rod.
MnO2 +
4 H+ + 2e–
![]() Mn2+
+ 2H2O
Mn2+
+ 2H2O
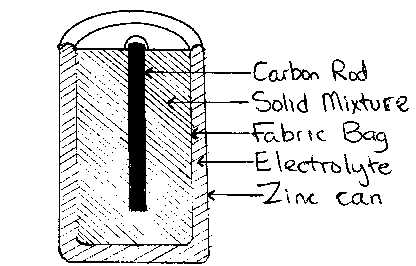
One disadvantage of this arrangement is that the zinc
container develops holes as the zinc dissolves and this allows the electrolyte
to leak out. An alternative design uses a pressed carbon outer container in a
steel can. Several strips of zinc are placed near the centre of the battery.
This ensures much more efficient use of the zinc and also makes the battery leak
proof.
These are essentially the same as carbon-zinc batteries
but use an alkaline solution of KOH as an electrolyte. Because the electrolyte
is not acidic, steel can be used as the positive terminal instead of carbon and
the whole steel container can be sealed, making the battery leak proof. The
reaction which generate the electricity in an alkaline battery are
Zn + 4OH–
![]() Zn(OH)4
2– +
2e–
Zn(OH)4
2– +
2e–
MnO2
+ 2H2O + e–
![]() Mn(OH)3 + 2OH–
Mn(OH)3 + 2OH–
They are based on compounds of nickel and cadmium. The
electrolytes in these batteries are made by heating a layer of powdered nickel
with nickel gauze to produce a porous, flexible layer of the metal. The battery
is then assembled from three layers
i)
a
porous nickel sheet soaked in a solution of nickel salt
ii)
a
sheet of absorbent paper soaked in potassium hydroxide solution
iii)
a
porous nickel sheet soaked in a solution of cadmium salt
The
sheets are rolled into a tight cylinder and sealed in a steel container. This is
fully leak proof.
The
KOH reacts with the salts and precipitates insoluble hydroxides into the pores
of each metal plate; so one plate is filled with Ni(OH)2(s) and the
other with Cd(OH)2(s) .
The cell is then charged by passing an electric current through it. The Ni(OH)2(s)
plate is made the anode and oxidation occurs at this electrode.
Ni(OH)2(s)
+ 2OH–
![]() NiO2(s) +
2H2O + 2e–
NiO2(s) +
2H2O + 2e–
The
Cd(OH)2(s) plate is the cathode and reduction occurs at this
electrode
Cd(OH)2(s)
+ 2e–
![]() Cd(s)
+ 2OH–
Cd(s)
+ 2OH–
The
electrolyte is not consumed, no gas is evolved and, because all the nickel and
cadmium compounds are solids, they remain trapped in the pores of the
electrodes.
When
the cell is used to generate electricity, the reverse reactions take place. The
NiO2 consumes electrons and so generates a positive charge. The Cd(s)
releases electrons and so generates a negative charge. In soluble
hydroxides are reformed in the pores of the electrodes.
The
operation of the cell can be represented by the equation
NiO2(s) +
Cd(s) +
2H2O


DISCHARGING
CHARGING
Ni(OH)2(s)
+ Cd(OH)2(s)
Several
hundreds of cycles of charging and discharging can be achieved giving a long
service life to the battery.
If
a clean iron nail is placed in normal tap water it rusts., but if a nail is
placed in freshly boiled air-free water in a sealed flask it does not rust. Also
if a nail is kept in dry air in a desiccator it too wont rust. Thus both air and
water together are necessary for the corrosion of iron.
Experimental
observations have shown that if the humidity of the air is less than 50%, no
corrosion occurs. If the humidity is above 80% the iron rusts rapidly.
Other
experiments have shown that an electrolyte is needed for corrosion to occur. So
NaCl solution (i.e. salt water) will accelerate corrosion.
Irregularities
in the surface exposed to the by the metal greatly influence the rate of
corrosion. Rusting is very much more for steel than for chemically pure iron. It
is also more rapid near imperfections in the surface and near areas of strain,
e.g. at the point and head of a nail.
The
water film on the surface of the iron is exposed to the atmosphere and will
dissolve any soluble substances that may be present. Thus, carbon dioxide in the
atmosphere will dissolve forming carbonic acid, and near the sea other
electrolytes. Such as carbon dioxide may be dissolved in the water film. These
dissolved substances will make the solution an electrolytic conductor.

The
presence of electrolytes in the solution could allow a current producing cell to
be set up between the iron as one electrode and an area of impurity, such as a
small crystal of carbon, as the other electrode.
A
cell can be set in the laboratory to examine the behaviour of iron under these
conditions
The
production of electrons by iron would involve dissolution of the iron
Fe
![]() Fe2+ + 2e–
oxidation
Fe2+ + 2e–
oxidation
The
electrons are consumed by a reduction process at the carbon electrode and this
accounts for the consumption of oxygen gas.
2H2O +
O2 + 4e–
![]() 4OH– reduction
4OH– reduction
So
the corrosion of iron is probably due to the transfer of electrons through the
metal to areas of impurity
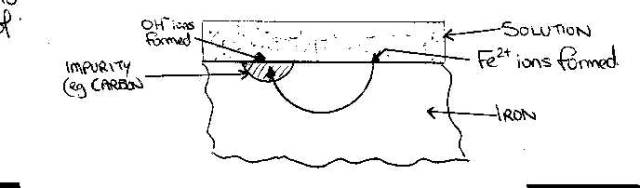
The
areas of impurity are areas to which the electrons drift, because electrons are
not being produced at these positions. Thus the oxidation of the iron to the
iron (II) state occurs with simultaneous formation of hydroxide ions.
The
result of obtaining a solution containing these ions can be seen by adding
sodium hydroxide to a solution of an iron (II) salt. A green gelatinous
precipitated of iron (II) hydroxide forms.
Fe2+
+ 4OH–
![]() Fe(OH)
2
Fe(OH)
2
On
standing, the green iron(II) hydroxide is slowly oxidized to brown iron(III)
hydroxide where it is in contact with air .
4Fe(OH)2
+ 2H2O
+ O2
![]() 4Fe(OH)3
4Fe(OH)3
iron
(III) hydroxide is hydrated iron (III) oxide
b.
the reasons why common methods of corrosion prevention are effective.
The
corrosion of iron can be greatly diminished or prevented in a number of ways.
i)
alloying
the iron with other elements
ii)
using
a protective coating
iii)
using
electrical protection
iv)
unreactive
metallic coatings
If chromium is alloyed with steel the product is called
stainless steel because of its resistance to corrosion. The addition of small
amounts of molybdenum further improves its resistance. Cast iron alloyed with
silicon is very resistant to corrosion but is weak structurally. However it is
useful in building chemical reaction vessels and other articles which require
resistance to attack by acids.
The most effective method of preventing corrosion of
underground steel structures is to completely coat them with an impervious
substance such as platicized coal / tar enamel.
Plastic coatings on metal structures are widely used to
prevent corrosion. Plastics in use include, polythene, polyvinylchloride (PVC),
epoxy resins, rubber and synthetic rubber. Steel can also be coated with glass.
These materials provide a barrier to corrosion under even the most severe
conditions.
Vitreous enamels and vinyl paints are used to protect car
bodies, refrigerators and washing machines.
Steel can be protected by being connected to a metal
higher than it on the activity series (or electrochemical series). Steel coated
with zinc is called galvanised iron and this method of protection is called
galvanic protection, or because the steel is protected by preferential corrosion
of the zinc, sacrificial protection. Even if the layer of zinc has small
imperfections the steel is still protected. The zinc dissolves in preference to
the iron because it forms positive ions more readily.
Zn
![]() Zn2+ + 2e–
Zn2+ + 2e–
The electrons are consumed on the iron, so preventing
corrosion.
Instead of using sacrificial protection, electrons can be
supplied by a D.C. generator. This method is used to protect steel wharves. The
steel wharf is wired to the negative terminal and the positive terminal is wired
to an anode. Only 4 to 5 volts is necessary and a current od a few milliamps per
square foot of structure is needed. The anode may be made up of large lumps of
steel such as engine blocks. In this case the scrap steel is slowly dissolved.
Fe
![]() Fe2+ + 2e–
Fe2+ + 2e–
The anode corrodes away where the electrical connection
is made. Silicon – iron anodes
are better v=because they corrode only very slowly. The latest development is to
use anodes made of titanium coated with a very thin layer of platinum, in which
case there is no corrosion of the anodes.
Steel is often protected from corrosion by using a thin
layer of tin. This is widely used in the manufacture of food containers, (the
tin is often coated with an impervious layer of lacquer). With tin plate it is
essential that the layers of tin has no imperfections, because tin is below iron
on the activity series (above iron on the electrochemical series).
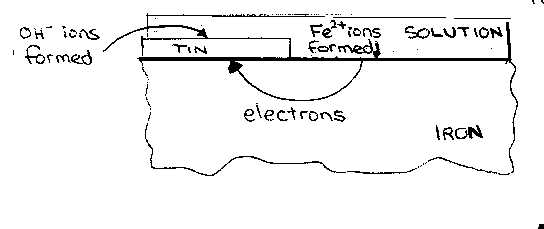
If there is a break in the tin plating, however small,
corrosion will commence on the exposed iron and will very rapid. The iron
dissolves because it forms positive ions more readily than tin.
Fe
![]() Fe2+ + 2e–
Fe2+ + 2e–
The electrons are consumed on the tin and so the iron
corrodes. For special purposes, steel is sometimes coated with gold or platinum
and it is essential that the layer has no imperfections because these metals are
very low on the activity series.